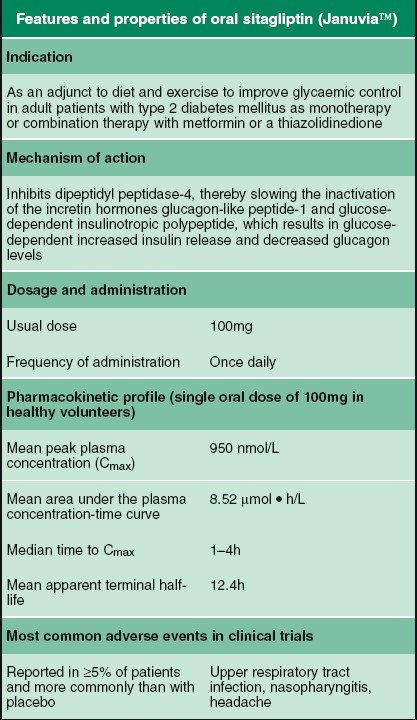
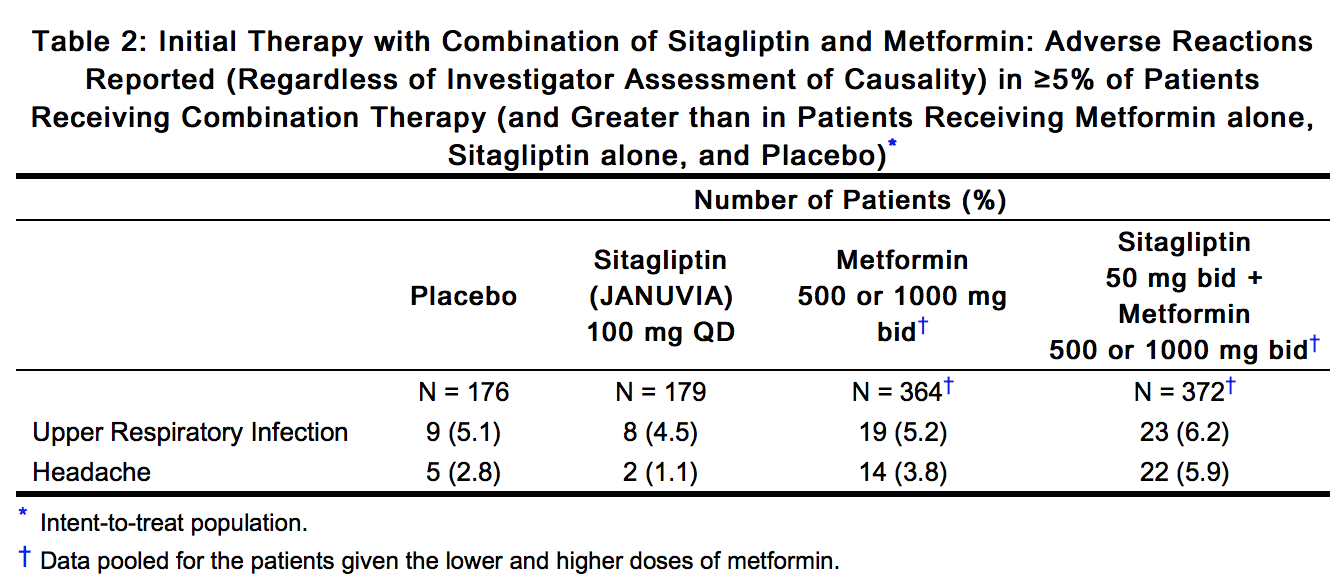
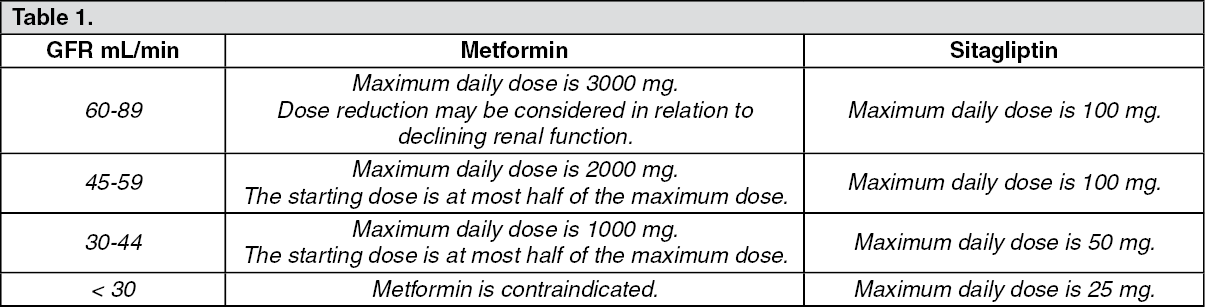
SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the Medical Product 1.1 Product Name: SITAPRIL M 50/500 (Sitagliptin 50 mg and Met
These highlights do not include all the information needed to use JANUVIA safely and effectively. See full prescribing information for JANUVIA. JANUVIA® (sitagliptin) tablets, for oral useInitial U.S. Approval: 2006

Pharmacokinetic–Pharmacodynamic Modelling of Biomarker Response to Sitagliptin in Healthy Volunteers - Kim - 2013 - Basic & Clinical Pharmacology & Toxicology - Wiley Online Library
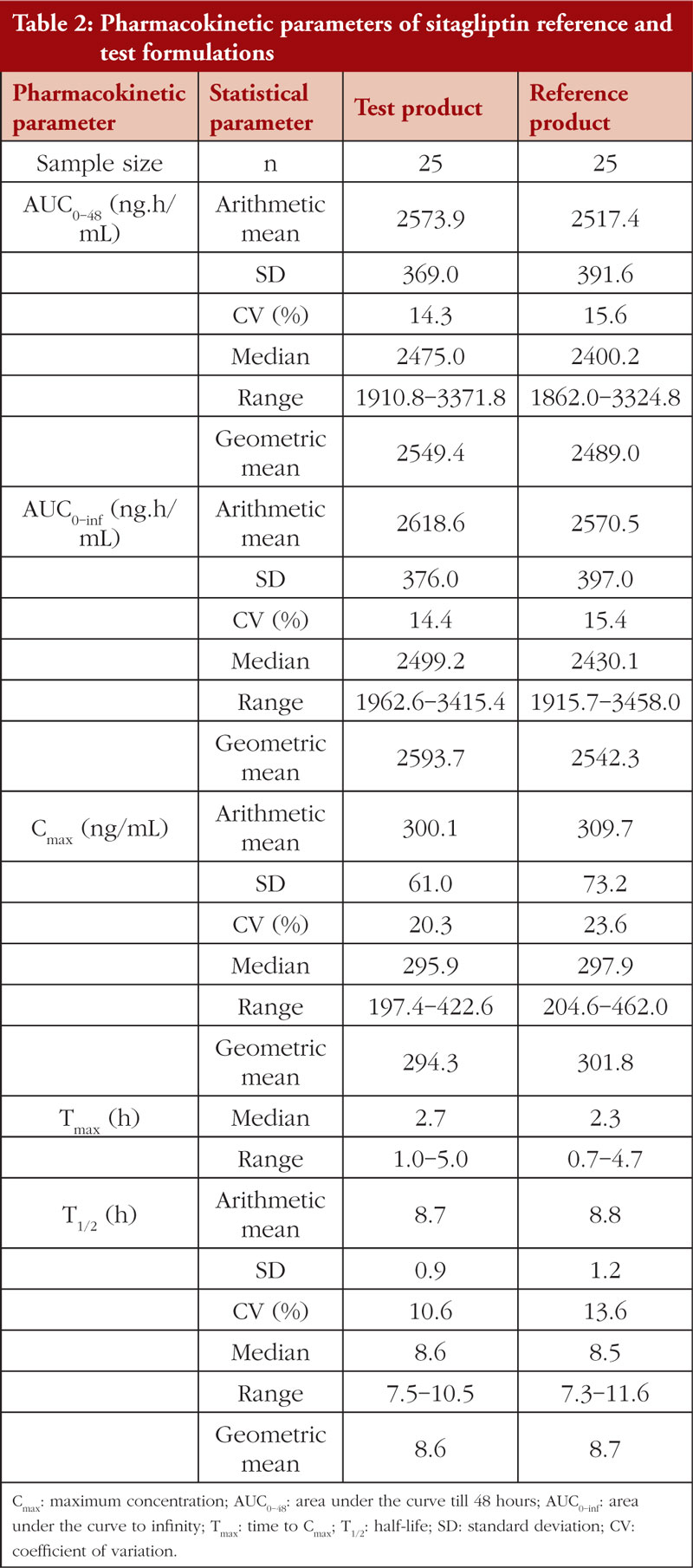

![PDF] Choosing a Gliptin | Semantic Scholar PDF] Choosing a Gliptin | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/afd0b84c9bc4712f65df36285a961e3af19ad967/5-Table2-1.png)